Systematic review of the economic evaluations of prevention strategies and screening interventions to reduce the risk of PTB in LMIC
Professor Simon Dixon
Dr Kinga Lowrie
On behalf of the PRIME collaborators.
School of Health and Related Research (ScHARR), University of Sheffield, United Kingdom.
23rd April 2019
Executive summary
The PRIME project is a programme of research looking at the effectiveness of interventions to improve child and maternal outcomes associated with preterm birth. Within the programme a discrete set of reviews has been included with the aim of informing stakeholder engagement programme and identifying future research questions relating to PTB.
The two reviews reported here related to economic evaluations of interventions that prevent PTB and economic evaluations of screening for diseases that are known to be associated with PTB. Both are limited to evaluations in low- and middle-income countries (LMICs). Searches were based on Medline (OvidSP), PsychInfo, and EMBASE.
The search resulted in 3,378 records, with 19 articles being finally included; seven economic evaluations of malaria prevention strategies, nine evaluations of syphilis screening, two alternative interventions to prevent PTB and one economic evaluation of alternative treatments for preterm labour.
The seven economic evaluations concerned with the prevention of malaria amongst pregnant women focused on intermittent preventative treatment in pregnancy (IPTp) with sulfadoxine–pyrimethamine (SP) with or without insecticide-treated bed nets (ITNs); variations to this included intermittent screening and treatment in pregnancy (ISTp), different dosing regimens for SP and artemether-lumefantrine (AL) as an alternative to SP. Overall, given differences in comparators, methods and general, it is difficult to produce any robust conclusions from the available economic evidence.
The nine studies of syphilis screening were all concerned with different types of tests. The majority of studies were based in sub-Saharan Africa, with two in China and the remaining studies covered countries in Latin America and Asia. There was one intervention for which there was reasonably robust evidence of cost-effectiveness across four studies - screening via rapid testing (without confirmatory testing) for antenatal syphilis. However, these studies do not assess whether non-rapid testing may be more cost-effective than rapid testing. The lack of other robust findings is due to a combination of poor methods, variability of methods and variability in the interventions/setting.
There were only two evaluations that considered other preventative strategies, one on group B streptococcal (GBS) immunization and one concerned with a quality improvement program in a health facility. The GBS study examines the potential cost-effectiveness of a vaccine that is in development, based on indicative costs and a range of efficacy estimates (Russell et al.,2017). The analysis shows it to be potentially cost-effective with the results being generalizable across a large number of countries. The study of a QI programme in Ghana also shows it to be cost-effective, however, this conclusion is undermined by the underlying study design; generalisability is also expected to be limited.
Only one study was found relating to treatment of preterm labour. This study is seriously flawed, although its underlying rationale is sound and highlights the need to undertake local cost-effectiveness analysis.
Looking across all the topics, five important methodological issues were identified that need addressing if future research is to be fruitful. When these issues are considered in the context of the wider PRIME research programme, the following eight recommendations for future research are highlighted:
- Future economic review topics need to be chosen in collaboration with stakeholders.
- When assessing future research, stakeholders need to assess whether the best approach is 'broad and simple' of 'narrow and complex' approach.
- When undertaken economic evaluations of PTB, postpartum costs and health outcomes need to be included. Identification of existing sources of these costs would be of value.
- When undertaken economic evaluations the WHO/WB threshold should not be used; a more appropriate threshold should be used. Consideration should be given as to the best threshold.
- Future research should be model-based and transparent so that other researchers are able to adapt the findings to their own setting.
- Any future research should give due consideration to the generalisability of findings so that the results can be used beyond the setting in which the research was undertaken.
- Further consideration should be given to the issue of including/excluding stillbirths from DALY/QALY estimates.
- Further research should consider the dual reporting of DALYs and QALYs.
1 Background
Preterm birth (PTB) is regarded as a delivery before 34 weeks’ gestation. It is a critical public health issue that accounts for 75% of neonatal mortalities with surviving infants having multiple health problems (Barros et al., 2010). Main causes of PTB include infections, cervical dysfunctions, or environmental and social factors (Goldenberg et al., 2008). The PRIME project is a programme of research looking at the effectiveness of interventions to improve child and maternal outcomes associated with preterm birth. The programme covers systematic literature reviews, stakeholder engagement, research prioritisation, guideline development and clinical research relating to possible determinants of preterm birth.
One important feature of developing and implementing interventions is their value for money, or cost-effectiveness. A separate stream of work relating to cost-effectiveness has not been included in PRIME, however, a discrete set of reviews has been included within the wider range of systematic reviews that are being undertaken within the project. The aim of these reviews is to inform the stakeholder engagement programme and identify important research questions that will support decision makers planning services and interventions relating to PTB. The first reviews are due to be completed by the end of April 2019.
Given the breadth of possible interventions that could be relevant to PRIME, ranging from primary prevention of pre-term birth, secondary prevention and postnatal care, we focused on three topics:
- Economic evaluations of interventions that prevent PTB or treat preterm labour*
- Economic evaluations of screening for diseases that are known to be associated with PTB
- Cost studies of PTB
The first two reviews provide information that is potentially relevant to research prioritisation and guideline development; they also provide examples of case studies that will help with the development of future evaluations. The third review is being undertaken to provide cost data for any future evaluations that PRIME may identify as being of particular importance. All three reviews are limited to evaluations in low- and middle-income countries (LMICs).
This document reports the results of the first two systematic reviews.
*The search included in the proposal did not include the treatment of preterm labour, however, this was included as it was felt that it was an important topic.
2 Why economic evaluation?
Economic evaluation is a comparative study of two or more interventions in terms of costs and outcomes (Drummond, 2005). Its primary focus is to assess efficiency by informing a decision to provide an intervention that helps maximise outcomes for a given budget. Just assessing costs cannot help with decisions about maximising outcomes. Just assessing one study cannot help with a choice.
Within health care, the outcome in question is usually ‘health’ itself. This can be captured by many measures, but when making choices across a budget that covers lots of different services, a generic measure of health is more useful; disability adjusted life years (DALYs) and quality adjusted life years (QALYs) are two such measures. DALYs and QALYs capture both morbidity and mortality within a single metric but do this in slightly different ways (which are beyond the scope of this paper).
To say that something is cost-effective means that providing it would generate more health than if the comparator were provided instead. In cases where one intervention is more effective and cheaper, this decision is simple. However, it is generally the case that the more effective intervention is more expensive, which means that if it is to be provided, money needs to be freed up elsewhere by stopping other interventions. In such circumstances we need to take into account those lost health benefits, too. If the more effective intervention is more cost-effective than the interventions being stopped to fund it, then health will be maximised by providing this new intervention. We make this assessment by comparing the incremental cost-effectiveness ratio (ICER) of the more expensive intervention against an indicative ICER for the interventions that would need to be stopped to fund it. This indicative ICER is often referred to as a 'threshol'; if we evaluate two treatments and the most effective treatment has an ICER that is below the threshold, we should fund it in order to maximise health, and vice versa. Crucially, the indicative ICER, or threshold, needs to approximate the cost-effectiveness of services currently funded by the health system (and which will be closed down to fund newer, more effective, treatments). Within the UK, this threshold has been estimate empirically, however, LMICs tend to use a threshold set by the World Health Organisation (WHO), which is equivalent to gross domestic product per capita (WHO, 2001). This is highly contentious, with many economists clearly demonstrating that this is much too high (Woods et al., 2016); this will have the effect of many analyses recommending interventions to be funded, when in fact, they should not be (if health is to be maximised). As such, use of the WHO threshold undermines the validity of many economic evaluations (and damages population health in those countries).
3 Methods
The systematic review of the published literature was conducted using the following methods.
Data sources
The following databases were searched in October 2018: Medline (OvidSP), PsychInfo, and EMBASE. No time restrictions were applied.
Studies were included if they were economic evaluations concerned with interventions to prevent pre-term birth in LMIC (including economic evaluations of screening strategies for diseases that lead to PTB). All types of economic evaluation designs were eligible for inclusion, including cost-effectiveness, cost-minimisation, and cost-benefit and cost-consequence analysis.
Studies that did not include outcome data, such as cost studies were excluded. Also, systematic reviews and the articles published in languages other than English were excluded. The review also did not include 'grey literature'.
Search terms
Search strategies adopted to each database included variations of the words "pre-term birth", "premature birth" and similar, combined with the search terms for economic evaluations and LMIC. The Boolean operators "OR" and "AND" were used to combine these terms. A detailed description of the search strategies is included in Appendix 1.
Study selection
Electronic search results from each of the databases were transferred to EndNote ver. 8, where the duplicates were removed. The titles and abstracts of each publication were then assessed for whether they met inclusion criteria. For the final set of records full versions were obtained. A second reviewer independently repeated the screening of the search results and assessed them for relevance. All inconsistencies were discussed.
4 Results
4.1 Studies identified
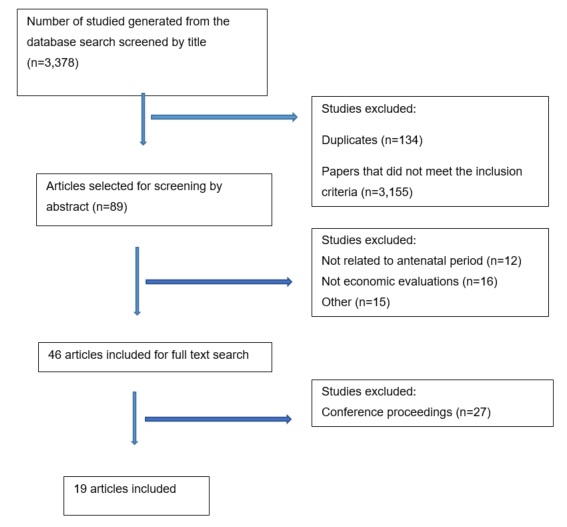
The search resulted in 3,378 records that were screened for relevance. 3,289 papers were excluded as they did not meet the inclusion criteria, whereas, 89 were selected to be screened by the abstract. As a result of the screening, a further 44 were excluded and 45 identified for full text screening. Finally, 19 articles were included. The process is presented in PRISMA diagram below.
The review identified seven economic evaluations of malaria prevention strategies, nine evaluations of syphilis screening, two alternative interventions to prevent PTB and one economic evaluation of alternative treatments for preterm labour. Five records relating to treatment with progesterone/corticosteroids were identified, but all were conference abstracts. One record relating to a systematic review and cost-effectiveness analysis of fetal fibronectin in Brazil, and another of nutrition interventions in LMICs, were identified, but both were limited to conference abstracts.
It should also be noted that several postnatal studies were excluded as they were dealing with a much broader population than preterm babies, for example, neonatal studies. It is recognised that such studies may be relevant to the care of preterm babies; whether such exclusions should be reconsidered is raised in the Discussion and Next Steps sections.
4.1.1 Malaria prevention
The seven economic evaluations concerned with the prevention of malaria amongst pregnant women in LMIC were all conducted in African countries (Table 1). The focus of these studies was on intermittent preventative treatment in pregnancy (IPTp) with sulfadoxine–pyrimethamine (SP) with or without insecticide-treated bed nets (ITNs); variations to this were intermittent screening and treatment in pregnancy (ISTp), different dosing regimens for SP and artemether-lumefantrine (AL) as an alternative to SP. One study evaluated cost-effectiveness of SP in comparison to cotrimoxazole (CTX) for HIV-positive women.
The three studies that examined the effect of ITNs in addition to chemoprophylaxis gave mixed results (Table 1, rows 2-4). The studies showed that in comparison with standard care ITNs are effective with ICERs ranging from US$17.22 per DALY averted (Becker-Dreps et al., 2009) to US$ 1.02 per DALY averted (Sicuri et al., 2010), which indicate that they are highly cost-effective. However, another suggested they were not as effective and more costly (Hansen et al., 2012). Transmission rate was identified as an important issue, with areas of low transmission showing prevention strategies to be less cost-effectiveness.
Four studies included in this review considered the cost-effectiveness of different drug regimens (Table 1, rows 5-8). One study examined different doses of SP e.g. two doses overall vs. monthly doses (Fernandes et al., 2015), another looked at screening and treatment with AL (Fernandes et al., 2016), another looked at alternative medication for HIV positive women e.g. CTX (Choi et al., 2017), and one study looked at service delivery by community or health centre staff (Mbonye et al., 2008b).
The results suggest that monthly doses of SP are more effective than restricting treatment to two doses only (Fernandes et al., 2015). Moreover, delivery of medication via community care was found more effective than when pregnant women were given the medication in health centers (Mbonye et al., 2008a). CTX appeared to be more cost-effective than IPTp-SP in women with HIV (Choi et al., 2017).
Methodological details of the studies are summarised in Table 2. All studies were based on high quality clinical evidence - generally, either RCTs or meta-analyses - however, the underlying studies have not been critically appraised. The studies on malaria prevention typically used the same outcome measures - the number of low birth weight deliveries, the number of maternal anemia and malaria cases – which were converted into DALYs. The method of conversion into DALYs varied as did the life expectancies used. No studies used quality adjusted life years as a method of valuing health outcomes. A cost-effectiveness threshold was specified in all studies and were based on the WB/WHO methodology that uses GDP per capita as a benchmark. On study used an additional, alternative, benchmark which they considered to be contextually more relevant (Becker-Dreps et al., 2009).
Costing took the societal perspective in four studies and a narrower, health care provider perspective in the remaining three studies. Whilst details of the costing methods were not always clear, it appears that all studies did not cost infant care beyond delivery, which will underestimate the need for more intensive care associated with LBW and ongoing care for many of those children. Four studies were based on cost-effectiveness models, with the remaining three studies undertaken in parallel with randomised controlled trials. Evaluations undertaken within the framework of an economic model are generally preferred as they allow easier adaptation by other researchers and hence use in other settings.
Overall, given differences in comparators, methods and general parameterisation (including the effectiveness of the underlying interventions), it is difficult to produce any robust conclusions from the available economic evidence. Two further problems are of note. First, the costs of infant care beyond delivery have been excluded in all evaluations to date. Evidence from the UK suggests that costs associated with low birth weight can be significant up to 10 years after birth (Petrou et al., 2009). Even if these costs are not representative of costs in LMICs, they point to a potentially important omission from the evaluations undertaken to date. Second, the cost-effectiveness thresholds have been based on those suggested by the WB/WHO, which have been heavily criticised and which have now been (partially) withdrawn by the WHO (Bertram et al., 2016).
In the absence of consistent and robust evidence from the published studies, if policy makers wish to use these studies to guide decisions in their own countries, they will need to identify which evaluation addresses the decision making problem that they face (e.g. comparators) and adapt the analysis to suit their own circumstances. Only in this way will the methods and data sources meet their requirements. Such work has been undertaken in the context of LMICs (Alshreef et al., 2019), however, it can be resource intensive.
Table 1: Summary of malaria prevention studies
Author |
Intervention |
Countries |
Outcome measure for CEA |
Results |
Reported conclusion |
(Becker- Dreps et al., 2009) |
ITNs in addition to usual care vs usual care alone |
Democratic Republic of Congo |
DALYs |
In comparison with standard care, ITNs yielded ICER US$17.22 per DALY averted, US$15.70 per LY saved and US$411 per infant death averted |
ITN distribution is a cost-effective addition to antenatal services. |
(Hansen et al., 2012) |
IPTp-SP given twice during pregnancy vs. ITNs alone vs. ITNs plus IPTp- SP |
Uganda |
DALYs |
ICERs for replacing IPTp-SP were (ii) 54 US$ per DALY averted and (iii) –53US$ per DALY averted. |
No evidence of economic grounds for replacing IPT-SP by ITNs alone or by a combined intervention in a setting of low and unstable transmission. |
(Sicuri et al., 2010) |
IPTp-SP + ITN vs. ITN
alone. |
Mozambique |
DALYs |
The ICER for maternal malaria was 41.46 US$ per DALY averted. The ICER including both the effect on the mother and on the newborn was 1.02 US$ per DALY averted. |
IPTp-SP in the context of ITNs is highly cost-effective if administered through existing antenatal services. |
(Fernandes et al., 2015) |
IPTp-SP with three or more doses(3+) vs two doses (2) |
Burkina Faso, Kenya, Malawi, Mali, Tanzania, Zambia |
DALYs |
In comparison with 2 doses of SP, monthly doses produced ICER 7.28$ per DALY averted. |
IPTp-SP3+ can be a highly cost- effective intervention. |
(Fernandes et al., 2016) |
ISTp-AL vs. IPTp-SP |
West Africa |
DALYs |
ICER -175.12 US$/DALY averted |
In settings with reduced IPTp efficacy, IST-AL has the potential to be cost-effective. |
(Choi et al., 2017) |
CTX vs IPTp-SP |
Ghana, Malawi, Kenya, Mozambique, Tanzania |
DALYs |
In all simulated countries, CTX was the preferred strategy, with ICER ranging from cost saving to 3.9 US$ per DALY averted. |
Daily CTX for HIN-infected pregnant women regardless of CD4 count is cost-effective. |
(Mbonye et al., 2008b) |
IPTp delivered via community center vs. IPTp delivered at health center |
Uganda |
DALYs |
The ICER of the community delivery system was US$1.1 per DALY averted. |
Community based delivery is cost- effective. |
Table 2: Methodological details of the malaria prevention studies
Author |
Design |
Time horizon |
Perspective |
Cost components |
Currency and price level |
Source of effectiveness data |
Cost-effectiveness threshold |
(Becker- Dreps et al., 2009) |
Decision tree |
Costs unclear. DALYs until death. |
Health care provider |
Cost of the ITNs, cost of transport of medication to medical centre, storage of ITNs, staff training, and staff wages. |
$US 2005 |
Prospective case-control study |
1xGDP per capita (WHO 2003) and 1xhealthcare expenditure per capita (assumption) |
(Sicuri et al., 2010) |
RCT |
Costs until birth.
DALYs until death. |
Societal |
Healthcare costs: nurses time, medication cost, cost of hospital admissions, cost of outpatient visits.
Patients’ costs: cost of travel to hospital, food and expenses. |
$US 2007 |
RCT |
World Bank, 1993 |
(Hansen et al., 2012) |
RCT |
Costs until birth DALYs until death |
Health care provider |
Cost of medical staff, cost of administering SP and bed nets, (IPTp is free), cost of outpatient visits for malaria treatment. |
$US 2004 |
RCT |
World Bank, 1993. |
(Fernandes et al., 2015) |
Decision tree |
Costs until birth.
DALYs until death. |
Societal |
Healthcare costs: Healthcare staff cost, and medication cost.
Household costs: transport, time spend travelling and waiting at the health facility. |
$US 2012 |
Published meta- analyses |
WHO, 2004 |
(Fernandes et al., 2016) |
Decision tree |
Costs unclear. DALYs until death. |
Health care provider |
Healthcare staff cost, medication cost, cost of hospitalisations and cost of outpatient visits due to malaria. |
$US 2012 |
RCT |
WHO, 1996 |
(Choi et al., 2017) |
Individual simulation model |
Costs until birth.
DALYs until death. |
Societal |
Healthcare costs:
Cost of medication and cost of healthcare staff.
Household costs:
Cost of antenatal visits, transportation costs and patient time. |
$US 2005 |
Published meta- analyses |
WHO, 2002 |
(Mbonye et al., 2008b) |
RCT |
Costs until birth.
DALYs until death. |
Societal |
Healthcare system costs: Cost of medication, and cost of its delivery. Patients’ costs: travel time and cost. |
Uganda shillings 2002/3 |
RCT |
World Bank, 1993. |
4.1.2 Syphilis screening
The second category of included studies were economic evaluations of syphilis screening strategies which were concerned with different tests. The majority of studies were based in sub-Saharan Africa, with two in China and the remaining studies covered countries in Latin America and Asia. Two studies examined syphilis screening in tandem with HIV testing (Bristow et al., 2016, Owusu-Edusei et al., 2014), two studies examined rapid tests alone compared to rapid plus confirmatory testing (Rydzak & Goldie, 2008), four studies compared rapid testing versus no screening (Kuznik et al., 2013; Terris-Prestholt et al., 2003) and one study compared confirmatory testing versus no screening (Hong et al., 2010). The underlying diagnostic performance data seems to be of reasonable quality; however, they have not been critically appraised. The included studies are summarised in Table 3.
The two studies that examined dual HIV/syphilis screening found it to be cost-effective. In one case (Bristow et al., 2016) this was based on dual screening being less costly and more effective, whilst in the other study, the conclusion was based on comparisons with other child health programmes, as opposed to any formal threshold (Owusu-Edusei et al., 2014). Both studies included neonatal costs, but not longer-term costs.
The two studies that looked at rapid test with and without confirmatory testing gave similar results with ICS being less costly and more effective than other options (Owusu-Edusei et al., 2011, Rydzak and Goldie, 2008). Both studies included neonatal costs, but not longer-term costs.
The four studies that looked at rapid testing versus no testing found testing to be more costly, but more effective; all concluded that testing was cost-effective (Kuznik et al., 2013, Kuznik et al., 2015, Schackman et al., 2007, Terris-Prestholt et al., 2003). These studies covered RPR and ICS tests, although these two types of test were not compared directly. The conclusions for these studies were based on WHO/WB thresholds and informal comparisons with other comparable programmes. None of these studies included costs beyond the immediate treatment period and so are likely to underestimate the cost-effectiveness of testing. Together with the very lower ICERs produced, this is highly suggestive that these interventions are cost-effective.
The single study that examined non-rapid testing together with confirmatory testing versus no screening produced an ICER of US$215 (Hong et al., 2010). The authors concluded that this was cost-effective but provided no threshold or any other basis on which to base this. In terms of methodology (Table 4), all studies were based on decision analytic modelling although varying structures were apparent. Four studies took the societal perspective. Only two studies relied on the use of WHO/WB thresholds to assess cost-effectiveness, with three using dominance to support conclusions of cost-effectiveness, thee using informal benchmarking and one providing no rationale whatsoever.
As with the malaria studies, none of the papers included long-term costs associated with congenital syphilis, although four included neonatal costs and one included treatment of congenital syphilis up to one year postpartum (Schackman et al., 2007). None of the studies included the costs and health benefits of preventing onward transmission of syphilis after successful treatment. Studies generally didn’t include reinfection during pregnancy, although this isn’t clear in many of the papers. Details were also lacking in many of the studies in relation to the precise treatment regimen for syphilis and the timing of the syphilis (and HIV) tests, which limits our ability to assess their relevance to other health care settings.
One area of methodology raised by two of the studies is whether stillbirths should be included within the DALY calculation (Rydzak and Goldie, 2008, Terris-Prestholt et al., 2003). Whilst life-years and DALYs lost are universally included for neonatal deaths, there is some disagreement about whether stillbirths should be as life expectancy is generally considered to start at the point of live birth (AbouZahr and Vaughan, 2000).
Once again, the variability in methods, data sources, comparators and setting make any firm conclusions open to criticism. However, the results suggest that rapid syphilis testing, with either ICS or RPR, compared to no testing, can be cost-effective; the favorable results even in the absence of other potential cost savings are promising. However, this is not to say that the results are generalisable, nor that rapid testing is more cost-effective than traditional testing.
Table 3: Summary of the syphilis screening studies
Author |
Intervention |
Countries |
Outcome measure for CEA |
Results |
Reported conclusion |
(Bristow et al., 2016) |
HIV rapid test vs. HIV and syphilis RPR test vs. single rapid test for HIV and syphilis vs. HIV rapid
and RPR/TPPA test. |
Malawi |
DALYs |
Dual HIV and syphilis rapid test strategy less costly and more DALYs averted than all the other strategies. |
The dual HIV and syphilis rapid test algorithm was the most cost- effective strategy. |
(Owusu- Edusei et al., 2014) |
No screening vs. screen for HIV only vs.
RPR/TPHA test for syphilis only vs. screen for both HIV and syphilis. |
China |
DALY |
Cost per DALY averted $168 for syphilis, $358 for HIV and syphilis, $536 for HIV. Adding syphilis screen to an existing HIV strategy 140US$ per DALY averted. |
Prenatal HIV screening programs that also include syphilis screening are likely to be more cost-effective than HIV screening alone. |
(Owusu- Edusei et al., 2011) |
Dual point-of-care test vs. ICS vs. RPR vs.
RPR/TPHA vs no screen |
Sub-Saharan Africa |
DALYs |
All screening options dominate no screening. ICS dominates all other screening options. |
The dual-POC test may help save cost in resource-poor settings where disease prevalence (and loss to follow-up) is high. |
(Rydzak and Goldie, 2008) |
RPR/TPHA vs. RPR vs.
ICS vs. no screening |
Sub-Saharan Africa |
Life-years (stillbirths and neonatal deaths) |
Rapid ICS screening dominates all other options. |
Universal prenatal syphilis screening using rapid point-of-care tests is cost-effective. |
(Kuznik et al., 2013) |
ICS syphilis screening vs. no screening. |
Sub-Saharan Africa |
DALYs |
The average cost per DALY averted was US$11 (range 2$US to US$48) |
Use of ICS tests for antenatal syphilis is highly cost-effective in SSA. |
(Kuznik et al., 2015) |
ICS syphilis screening vs. no screening. |
30 countries in Asia, South and Central America* |
DALYs |
ICER of syphilis screening is 53US$ (range US$10- US$332) in Asia and 60US$ (range US$5-225US$) in Latin America |
Antenatal syphilis screening is highly cost-effective in all the 11 Asian and 20 Latin American countries assessed. |
(Schackman et al., 2007) |
No screening (rural) or RPR test with 1 wk follow- up (urban) vs. RPR rapid test |
Haiti |
DALYs averted |
Rapid testing with immediate treatment has a cost- effectiveness ratio of $6.83/DALY in rural settings and
$9.95/DALY in urban settings. |
Integrating a new rapid syphilis test into prenatal care and HIVE testing is cost-effective. |
(Terris- Prestholt et al., 2003) |
RPR test vs. no screening |
Tanzania |
DALYs averted |
Rapid syphilis screening has an ICER of 10.56US$ per DALY saved (incl. stillbirths). |
On-site syphilis screening and treatment are extremely good value for money. |
(Hong et al., 2010) |
TRUST/TPPA screening vs. no screening |
China |
DALYs |
ICER of 215 US$ per DALY averted |
It can be concluded that this program is cost-effective. |
* Bangladesh, Cambodia, China, India, Indonesia, Laos, Malaysia, Philippines, Thailand, Vietnam, Argentina, Belize, Bolivia, Brazil, Chile, Colombia, Costa Rica, Cuba ,Dominican Republic, Ecuador, El Salvador, Guatemala, Haiti, Honduras, Mexico, Nicaragua, Paraguay, Peru, Uruguay, Venezuela.
Table 4: Methodological details of the syphilis screening studies
Author |
Design |
Time horizon |
Perspective |
Cost components |
Currency and price level |
Source of effectiveness data |
Cost-effectiveness threshold |
(Bristow et al., 2016) |
Decision tree |
Costs up to one year postpartum. DALYs until death. |
Societal |
Healthcare costs: Test, staff time, neonatal costs, cost of HIV and syphilis treatment.
Patient costs: travel and time. |
2012 $US |
Test performance and treatment effectiveness from multiple literature sources. |
None. Conclusion based on strong dominance. |
(Owusu- Edusei et al., 2014) |
Markov cohort model |
Costs unclear but includes neonatal period. Life years until death. |
Healthcare provider |
Cost of tests, cost of syphilis treatments, cost of HIV treatment, neonatal treatment |
$US 2010 |
Test performance and treatment effectiveness from multiple literature sources. |
None. Conclusion based on comparisons between the four study comparators. |
(Owusu- Edusei et al., 2011) |
Markov cohort model |
Costs unclear but includes neonatal period. DALYs until death. |
Societal and healthcare provider |
Healthcare costs: test, counselling, treatment and neonatal costs Patient costs: time and travel cost |
2008 $US |
Test performance and treatment effectiveness from multiple literature sources. |
None. Conclusion based on comparisons between the four study comparators. |
(Rydzak and Goldie, 2008) |
Markov cohort model |
Costs unclear but includes neonatal period. Life years until death. |
Healthcare provider |
Cost of tests, medical staff, maternal treatment, neonatal treatment |
$US 2004 |
Test performance and treatment effectiveness from multiple literature sources. |
World Bank (2006) and WHO (2001) |
(Kuznik et al., 2013) |
Unclear |
Costs unclear but includes neonatal period. Life years until death. |
Healthcare provider |
Cost of test, staff and medication |
$US 2011 |
Test performance from systematic review.
Treatment effectiveness from meta-analysis. |
None. Vague benchmarking to UK practice and per capita income. |
(Kuznik et al., 2015) |
Unclear |
Costs until end of treatment. DALYs until death. |
Healthcare provider |
Cost of test, staff and medication |
$US 2012 |
Test performance from systematic review.
Treatment effectiveness from another study. |
WHO (2011) |
(Schackma n et al., 2007) |
Decision tree |
Costs up to 1 year postpartum. DALYs until death. |
Societal |
Healthcare costs: medical staff, tests Patient costs: treatment, patient travel |
$US 2004 |
Test performance from diagnostic accuracy study. Treatment effectiveness from historical data. |
World Bank 1993 and WHO 2001 |
(Terris- Prestholt et al., 2003) |
Unspecified model |
Costs up to end of treatment. DALYs until death. |
Healthcare provider |
Capital, medical staff, tests and treatment |
$US 2001 |
Test performance and treatment effectiveness from observational data. |
No explicit threshold used. Comparisons made against ICERs for comparable programmes. |
(Hong et al., 2010) |
Unclear |
Costs until end of treatment. DALYs until death. |
Societal |
Healthcare costs: Screening Patients costs: medical expenses, loss of income, travel costs |
2005 $US |
Test performance and treatment effectiveness from multiple literature sources. |
None. |
4.1.3 Other interventions for the prevention of PTB
There were only two evaluations that considered other preventative strategies, one on group B streptococcal (GBS) immunization and one concerned with a quality improvement program in a health facility (Table 5).
GBS infections are a leading cause of neonatal sepsis and meningitis, that in western countries are easily prevented by using antibiotics during labour and appropriate testing (Edmond et al., 2012). As these are not always available in LMIC, immunization might be an option if an appropriate vaccine can be developed; a pentavalent GBS vaccine is currently in development. The study identified in this review examines the potential cost-effectiveness of the vaccine based on indicative costs and a range of efficacy estimates (Russell et al., 2017). In comparison with the other studies reviewed to date, this study provides a more detailed evaluation, albeit of a hypothetical intervention. Amongst the methods employed, two interesting features are of note (Table 6). Firstly, 37 countries are statistically assigned to four clusters, with the cost-effectiveness analysis then being undertaken in one country in each cluster; this approach aims to provide generalizable results whilst keeping local data requirements and analytic workload to a minimum. Secondly, the authors highlight the recent debate relating to the inadequacy of the WHO cost-effectiveness threshold of 1 x GDP per capita, so in addition to using this usual threshold, the use 0.5 x GDP per capita which has a more robust empirical basis (although this too is potentially flawed).
The second study evaluated a 5-year quality improvement programme in a health facility in Ghana, that addressed several aspects of care delivery including optimising services, and personnel training and quality improvement. As such, the intervention probably spans prevention of preterm birth, treatment of preterm labour and care of the baby. Main outcomes considered included maternal deaths and stillbirths which were converted into DALYs. The evaluation used an observational study, with a before-after design from a single centre to estimate effectiveness; no model was developed for this. As such, the value of its results are open to question as the estimate of effectiveness is open to biases, they are unlikely to be generalizable and the analyses are hidden from view (and as such, they cannot be adapted to other settings). The threshold by which cost-effectiveness was measured was the 1xGDP per capita WHO threshold, which has been strongly criticised, however, the ICER was around one-tenth of that which provides some reassurance about the conclusion.
Overall, the analysis of the cost-effectiveness of a planned GBS vaccine shows it to be potentially cost-effective. However, whilst the model is clearly described and relatively well detailed, further developments will be required once data are available on the vaccine. The use of clustering to inform generalisability and the use of a more empirically robust cost-effectiveness threshold examples of good practice. The study of a quality improvement programme in Ghana also shows it to be cost-effective, however, this conclusion is undermined by the underlying study design; generalisability is also expected to be limited.
Table 5: Summary of the studies investigating other interventions
Author |
Intervention |
Countries |
Outcome measure for CEA |
Results |
Reported conclusion |
(Russell et al., 2017) |
GBS vaccine vs no immunization |
Guinea-Bissau, Uganda, Nigeria and Ghana representing 4 clusters
covering 37 countries |
DALYs |
At a vaccination cost of $7/dose, maternal immunization would create $320-$350/DALY averted in Guinea-Bissau, Nigeria and Ghana. In Uganda immunization would be US$572 per DALY averted. |
Maternal GBS immunization could be a cost-effective intervention in low-income sub-Saharan Africa. |
(Goodman et al., 2017) |
Quality improvement programme |
Ghana |
DALYs |
ICER of $158 per DALY averted, with a range from sensitivity analysis of $97-$218. |
Quality improvement interventions to reduce maternal and fetal mortality in low resource settings can be highly cost effective. |
Table 6: Methodological details of the studies investigating other interventions
Author |
Design |
Time horizon |
Perspective |
Cost components |
Currency and price level |
Source of effectiveness data |
Cost-effectiveness threshold |
(Russell et al., 2017) |
Decision tree with embedded Markov nodes |
Costs up to 1 year postpartum. DALYs until death. |
Health care provider |
Vaccine price and delivery, infant GBS treatment cost |
2014 $US |
Expert opinion |
WHO (1xGDP per capita) and Woods et al (0.5 GDP per capita) |
(Goodman et al., 2017) |
Observational study |
Costs unclear, but probably up to discharge. DALYs until death. |
Health care provider |
Costs of the QI programme (e.g. training and equipment) |
2015 $US |
Observational study |
WHO (1xGDP per capita) |
4.1.4 Treatment of preterm labour
Only one study was found relating to treatment of preterm labour (Jakovljevic et al., 2008). The rationale for the study is sound, with the authors highlighting that treatments deemed cost-effective in high-income countries (HICs) were not used in Serbia and Montenegro and this may be due to different cost structures; Serbia and Montenegro has low hospital costs, but faces similar drug costs to HICs.
The study has a simple methodology, with the primary economic outcome measure being pregnancy weeks (Table 7). With no external cost-effectiveness threshold, the conclusion is about the relative cost-effectiveness of the two drugs; we do not know whether either is cost-effective in absolute terms.
The evaluation used a non-randomised study, from a single centre to estimate effectiveness; no model was developed for this. As such, the value of its results are open to question as the estimate of effectiveness is open to biases, they are unlikely to be generalizable and the analyses are hidden from view (and as such, they cannot be adapted to other settings). Neither costs nor outcomes are measured after birth for the primary analysis, so the consequences of preterm birth are not captured. Maternal quality of life was captured after birth using a patient reported outcome measure, but this was not reported.
Overall, this study is seriously flawed, although its underlying rationale is sound and highlights the need to undertake local cost-effectiveness analysis.
Table 7: Summary of the studies investigating the treatment of preterm labour
Author |
Intervention |
Countries |
Outcome measure for CEA |
Results |
Reported conclusion |
(Jakovljevic et al., 2008) |
Ritodrine and fenoterol for the treatment of preterm labour |
Serbia and Montenegro |
Pregnancy weeks |
The cost of treatment per week of pregnancy prolongation was 3345 Serbian Dinars in the fenoterol group and 4181 Serbian Dinars in the ritodrine group (p>0.05) |
Fenoterol treatment tends to be more cost-effective and its lower acquisition cost is an advantage to this treatment option. |
Table 8: Methodological details of the studies investigating the treatment of preterm labour
Author |
Design |
Time horizon |
Perspective |
Cost components |
Currency and price level |
Source of effectiveness data |
Cost-effectiveness threshold |
(Jakovljevic et al., 2008) |
Non- randomised trial |
Costs up to delivery. Outcomes up to delivery. |
Payer |
Drugs, tests, consumable, hospital stay. |
Unclear, but probably 2006 Serbian Dinars |
Non-randomised trial. |
None. Conclusion based on comparisons between the two study comparators. |
5 Discussion
5.1 Summary
This review synthesizes the evidence on the cost-effectiveness of strategies for the prevention of PTB or treatment of preterm labour in LMIC countries. We found nineteen economic evaluations on this topic. Seven economic evaluations studied the prevention of malaria amongst pregnant women using intermittent preventative treatment in pregnancy (IPTp) with sulfadoxine–pyrimethamine (SP) with or without insecticide-treated bed nets (ITNs). Nine studies examined the cost-effectiveness of syphilis screening strategies using various tests and testing procedures. Two evaluations considered other preventative strategies; one on group B streptococcal (GBS) immunization and one concerned with a quality improvement program in a health facility. Only one study was found relating to treatment of preterm labour, which evaluated the cost-effectiveness of ritodrine vs. fenoterol.
The number of studies relating to malaria prevention and screening for syphilis initially suggested that there would be sufficient evidence to generate useful policy recommendations. However, for the malaria studies very few of the studies compared the same treatment or testing regimen; different drugs were used in different settings and different patient populations. For the syphilis screening studies, the evaluations formed small sub-categories, within which there were two to four studies.
In addition, in the variability in comparators within each topic area, we identified other issues that limited our ability to draw firm conclusions. The main problems were the lack of any consideration of longer-term health outcomes and costs associated with preterm birth and the use of inappropriate cost-effectiveness thresholds. A further limitation was that the studies were largely confined to one country without any consideration of the generalisability of their results.
Taking these limitations together, we feel that only one finding is sufficiently robust to support; rapid testing with RPR or ICS for syphilis in pregnancy versus no testing appears to be cost-effective in LMICs cost-effective. However, this conclusion includes several subjective judgements about generalisability, and the impact of potential flaws in the underlying studies.
The other three studies need to be considered separately as they are evaluating very different interventions and involve different evidence based and analyses. The evaluation of a GBS vaccine is based on high quality analyses that produces generalisable results, however, the vaccine is hypothetical. As such, the analysis indicates that when such a vaccine is available, it is expected to be cost-effective. The evaluation of a quality improvement programme is weakened by the evidence of effectiveness, but its estimated cost-effectiveness suggests that it is worthy of consideration in similar settings. The study of ritodrine vs. fenoterol is based on poor quality evidence and should be ignored.
5.2 Strengths and weakness
The review is based on robust search methods and study identification, however, there are some limitations. Firstly, we have not undertaken a structured critical appraisal of the studies. The reason for this is that the sparsity and variability in the identified literature precludes an robust conclusions irrespective of the quality of the literature; as such, critical appraisal will not have a notable impact on our conclusions. The only exception to this is the topic of rapid testing with RPR or ICS for syphilis in pregnancy versus no testing, for which an in-depth structured critical appraisal is underway. As such, the review may be best referred to as a scoping review, rather than a systematic review.
Second, the formulation of the searches was around search terms that included ‘preterm birth’, however, it became clear in the sifting process that studies that may be relevant to the care of preterm babies may have been excluded as they referred to neonatal care or low birthweight infants, without any reference to preterm birth. It would be possible to reformulate the review to capture all studies related to prematurity, but this was not the intention of this study.
Thirdly, we did not undertake a grey literature search of cost-effectiveness literature. This decision will be revisited once the results to date have been discussed by the study collaborators; whilst the inclusion of grey literature would be welcomed, given the evolving priorities of the research group, examination of another topic may be considered more valuable.
5.3 Compare and contrast
It should be recognised that other reviews of economic evaluations are of relevance to PRIME, even though they are not specifically restricted to preterm birth. Of particular note are four studies undertaken by the PRICELESS SA group at the University of Witwatersrand, henceforth ‘Wits’ (Table 9). Given the different objectives of their work and ours, the overlap in literature is minimal. However, the Wits work demonstrates how systematic reviews, in tandem with policy formation and cost-effectiveness analysis can provide important information for prioritisation and investment planning.
Methodologically, Wits have undertaken simple modelling to address a broad topic area and many interventions. Our review identified more detailed cost-effectiveness analyses that are specific to a particular comparison, for example, ritodrine vs. fenoterol in the treatment of preterm labour. Any future economic modelling work within PRIME needs to consider which general approach - ‘broad and simple’ versus ‘narrow and complex’ - is most relevant to the aims of the programme.
Table 9: Systematic reviews and modelling studies undertaken by PRICELESS SA
Author and year |
Topic |
Results |
Conclusions |
(Nkonki et al., 20017) |
Community interventions to reduce child mortality. Nine interventions were evaluated (e.g. breastfeeding promotion) |
The nine interventions can prevent 8891 deaths by 2030. At scale, US$169.5 million per year will be required to deliver these interventions via community health workers. |
The use of community health workers offers enormous opportunities for saving lives. These programmes require appropriate financial investments. |
(Michalow et al., 2015) |
Prevention of stillbirths, maternal and newborn mortality. Thirteen interventions were evaluated (e.g. fetal growth restriction detection and management). |
Full coverage of the 13 interventions in 2030 could reduce the stillbirth rate by 30%, costing US$420 million annually. All interventions were highly cost- effective. |
Improved coverage of 13 interventions that are already recommended could significantly impact the rates of stillbirth and maternal and neonatal mortality. |
(Chola et al., 2015) |
Hospital interventions to prevent child and maternal deaths, (e.g. TB management in women). |
The fifteen interventions will save an additional 9,000 newborns and children and 1,000 mothers annually. An additional US$370 will be required. |
Focus on HIV and TB during early pregnancy is essential. Strategic gains could be realised by targeting vulnerable populations and districts with the worst health outcomes. |
(Maredza et al., 2016) |
Systematic review of interventions to reduce neonatal morbidity and mortality |
Twenty-seven economic evaluations were identified. Studies were of mixed quality, and in addition, comparing results between countries was difficult. |
Strengthening community care could be an important component of averting neonatal deaths which do not require the health system infrastructure of hospital interventions. |
5.4 Methodological challenges
Any systematic review of cost-effectiveness analyses will identify methodological problems in the literature it identified, and the body of work described here is no different. However, rather than obsess over generic technical issues such as sensitivity analysis, we have highlighted methodological issues that are of particular importance and relevance to the evaluation of preterm birth interventions in LMICs.
The majority of studies have used the WHO/WB thresholds for assessing cost-effectiveness. As described earlier, this will have the effect of many analyses recommending interventions to be funded, when in fact, they should not be, if health is to be maximised.
The majority of studies have excluded postpartum costs and all studies have excluded the long-term costs associated with preterm birth. This will have the effect of underestimating the cost-effectiveness of the most effective intervention.
The results of many studies are not generalisable to other studies due to a narrow evidence base or the lack of any consideration for cost-effectiveness in other settings. Likewise, many studies were not undertaken and presented in a way that allows other researcher to adapt the original findings so that they are more appropriate to an alternative setting. Evaluations undertaken within the framework of an economic model are generally preferred as they allow easier adaptation by other researchers and hence use in other settings.
All but two studies have used DALYs as the primary measure of outcome, with none using QALYs. The relative advantages of these two generic measures of health has been the subject of a long debate, however, we hold the view that the use of QALYs is preferred (for reasons beyond the scope of this paper).
Finally, we highlighted that there was disagreement about whether the years of life lost due to a still birth should be included within DALY (or QALY) estimates. Further consideration of this issue is needed.
5.5 Future research
This review provides a useful starting point and provides a useful resource for modelling to start. It has also provided us with evidence and inspiration to identify several future research topics to be considered by the PRIME collaborators.
- Future economic review topics need to be chosen in collaboration with stakeholders. The limitations of the reviews presented here have been reported – the lack of a grey literature search, a full critical appraisal of all studies and specific focus on preterm birth. Whether these should be addressed, or whether we should move onto another review, needs to be considered. Whether that next review should be the originally planned review – costs associated with PTB – or another, should also be considered.
- When assessing future research, stakeholders need to assess whether the best approach is ‘broad and simple’ of ‘narrow and complex’ approach.
- When undertaken economic evaluations of PTB, postpartum costs and health outcomes need to be included. Identification of existing sources of these costs would be of value.
- When undertaken economic evaluations the WHO/WB threshold should not be used; a more appropriate threshold should be used. Consideration should be given as to the best threshold.
- Future research should be model-based and transparent so that other researchers are able to adapt the findings to their own setting.
- Any future research should give due consideration to the generalisability of findings so that the results can be used beyond the setting in which the research was undertaken.
- Further consideration should be given to the issue of including/excluding stillbirths from DALY/QALY estimates.
- Further research should consider the dual reporting of DALYs and QALYs.
6 Conclusions
We undertook a systematic review of economic evaluations of interventions to prevent PTB in LMICs and identified 19 studies. The only intervention for which there was reasonably robust evidence of cost-effectiveness was screening via rapid testing (without confirmatory testing) for antenatal syphilis, however, these studies do not assess whether non-rapid testing may be more cost-effective than rapid testing. The lack of other robust findings is due to a combination of poor methods, variability of methods and variability in the interventions/setting. In addition, the review identified five important methodological issues that need addressing if future research is to be fruitful. Consequently, eight recommendations for the future research programme of PRIME were identified.
Appendix 1: Search terms
Search terms used in Ovid
1. ((preterm or pre-term or premature) adj3 (birth* or labo?r or deliver*)).tw. |
2. exp PREMATURE BIRTH/ |
3. exp OBSTETRIC LABOR, PREMATURE/ |
4. (low birth weight* or low birthweight*).tw. |
5. exp Perinatal Mortality/ |
6. exp Abortion, Spontaneous/ |
7. exp Fetal Death/ |
8. exp Stillbirth/ |
9. (still birth* or still-birth* or stillbirth* or pre-term birth* or pre term birth* or preterm birth* or f?etal death*).tw. |
10. 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 |
11. health economics.mp. [mp=hw, ab, ti, ct, ot, nm, fx, kf, px, rx, ui, sy] |
12. exp economic evaluation/ |
13. exp health care cost/ |
14. pharmacoeconomics/ or "drug cost"/ or drug utilization/ or "utilization review".mp. [mp=hw, ab, ti, ct, ot, nm, fx, kf, px, rx, ui, sy] |
15. socioeconomics/ and economics/ |
16. *socioeconomics/ |
17. Economic model.mp. [mp=hw, ab, ti, ct, ot, nm, fx, kf, px, rx, ui, sy] |
18. *fee/ |
19. *"cost"/ |
20. cost*.ti. |
21. (cost* adj2 (effective* or utilit* or benefit* or minimi* or evaluat* or analy* or study or studies or consequenc* or compar* or efficienc* or variable or unit or estimate* or variable* or unit)).ab. |
22. (price or prices or pricing).tw. |
23. (economic* or pharmacoeconomic* or pharmaco-economic*).tw. |
24. budget*.tw. |
25. (value adj1 (money or monetary)).tw. |
26. (financ* adj2 (cost* or data or "health care")).tw. |
27. financ*.tw. and economics/ |
28. (expenditure* not energy).tw. |
29. quality adjusted life year/ |
30. (eq-5d* or eq5d* or euroquol* or euroqol* or euroqual* or euro-quol* or euro-qol* or euro- qual*).tw. |
31. quality adjusted life.tw. |
32. (qaly or qalys or qald or qale or qtime).tw. |
33. disability adjusted life.tw. |
34. (daly or dalys).tw. |
35. (SF6D or sf 6d or short form 6d or shortform6d).tw. |
36. health* year* equivalent*.tw. |
37. (hye or hyes).tw. |
38. health utilit*.tw. |
39. (hui1 or hui2 or hui3).tw. |
40. disutil*.tw. |
41. standard gamble*.tw. |
42. (time trade off or time tradeoff).tw. |
43. (hqol or h qol or hr qol or hrqol).tw. |
44. (pqol or qls).tw. |
45. 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18 or 19 or 20 or 21 or 22 or 23 or 24 or 25 or 26 or 27 or 28 or 29 or 30 or 31 or 32 or 33 or 34 or 35 or 36 or 37 or 38 or 39 or 40 or 41 or 42 or 43 or 44 |
46. exp animals/ not (exp animals/ and exp humans/) |
47. exp veterinary medicine/ |
48. animal experiment/ |
49. ((energy or oxygen* or metaboli*) adj3 (expenditure* or cost*)).tw. |
50. 46 or 47 or 48 or 49 |
51. 45 not 50 |
52. Developing Countries.sh,kf. |
53. (Africa or Asia or Caribbean or West Indies or South America or Latin America or Central America).hw,kf,ti,ab,cp. |
54. (Afghanistan or Albania or Algeria or Angola or Antigua or Barbuda or Argentina or Armenia or Armenian or Aruba or Azerbaijan or Bahrain or Bangladesh or Barbados or Benin or Byelarus or Byelorussian or Belarus or Belorussian or Belorussia or Belize or Bhutan or Bolivia or Bosnia or Herzegovina or Hercegovina or Botswana or Brasil or Brazil or Bulgaria or Burkina Faso or Burkina Fasso or Upper Volta or Burundi or Urundi or Cambodia or Khmer Republic or Kampuchea or Cameroon or Cameroons or Cameron or Camerons or Cape Verde or Central African Republic or Chad or Chile or China or Colombia or Comoros or Comoro Islands or Comores or Mayotte or Congo or Zaire or Costa Rica or Cote d'Ivoire or Ivory Coast or Croatia or Cuba or Cyprus or Czechoslovakia or Czech Republic or Slovakia or Slovak Republic or Djibouti or French Somaliland or Dominica or Dominican Republic or East Timor or East Timur or Timor Leste or Ecuador or Egypt or United Arab Republic or El Salvador or Eritrea or Estonia or Ethiopia or Fiji or Gabon or Gabonese Republic or Gambia or Gaza or Georgia Republic or Georgian Republic or Ghana or Gold Coast or Greece or Grenada or Guatemala or Guinea or Guam or Guiana or Guyana or Haiti or Honduras or Hungary or India or Maldives or Indonesia or Iran or Iraq or Isle of Man or Jamaica or Jordan or Kazakhstan or Kazakh or Kenya or Kiribati or Korea or Kosovo or Kyrgyzstan or Kirghizia or Kyrgyz Republic or Kirghiz or Kirgizstan or Lao PDR or Laos or Latvia or Lebanon or Lesotho or Basutoland or Liberia or Libya or Lithuania or Macedonia or Madagascar or Malagasy Republic or Malaysia or Malaya or Malay or Sabah or Sarawak or Malawi or Nyasaland or Mali or Malta or Marshall Islands or Mauritania or Mauritius
or Agalega Islands or Mexico or Micronesia or Middle East or Moldova or Moldovia or Moldovian or Mongolia or Montenegro or Morocco or Ifni or Mozambique or Myanmar or Myanma or Burma |
or Namibia or Nepal or Netherlands Antilles or New Caledonia or Nicaragua or Niger or Nigeria or Northern Mariana Islands or Oman or Muscat or Pakistan or Palau or Palestine or Panama or Paraguay or Peru or Philippines or Philipines or Phillipines or Phillippines or Poland or Portugal or Puerto Rico or Romania or Rumania or Roumania or Russia or Russian or Rwanda or Ruanda or Saint Kitts or St Kitts or Nevis or Saint Lucia or St Lucia or Saint Vincent or St Vincent or Grenadines or Samoa or Samoan Islands or Navigator Island or Navigator Islands or Sao Tome or Saudi Arabia or Senegal or Serbia or Montenegro or Seychelles or Sierra Leone or Slovenia or Sri Lanka or Ceylon or Solomon Islands or Somalia or South Africa or Sudan or Suriname or Surinam or Swaziland or Syria or Tajikistan or Tadzhikistan or Tadjikistan or Tadzhik or Tanzania or Thailand or Togo or Togolese Republic or Tonga or Trinidad or Tobago or Tunisia or Turkey or Turkmenistan or Turkmen or Uganda or Ukraine or Uruguay or USSR or Soviet Union or Union of Soviet Socialist Republics or Uzbekistan or Uzbek or Vanuatu or New Hebrides or Venezuela or Vietnam or Viet Nam or West Bank or Yemen or Yugoslavia or Zambia or Zimbabwe or Rhodesia).hw,kf,ti,ab,cp.
55. ((developing or less* developed or under developed or underdeveloped or middle income or low* income or underserved or under served or deprived or poor*) adj (countr* or nation? or population? or world)).ti,ab.
56. (low* adj (gdp or gnp or gross domestic or gross national)).ti,ab.
57. (low adj3 middle adj3 countr*).ti,ab.
58. (lmic or lmics or third world or lami countr*).ti,ab.
59. transitional countr*.ti,ab.
60. 52 or 53 or 54 or 55 or 56 or 57 or 58 or 59
61. 10 and 51 and 60
Search terms used in Medline
1. (still birth* or still-birth* or stillbirth* or pre-term birth* or pre term birth* or preterm birth* or f?etal death*).tw. |
2. exp Stillbirth/ |
3. exp Fetal Death/ |
4. exp Abortion, Spontaneous/ |
5. exp Perinatal Mortality/ |
6. (low birth weight* or low birthweight*).tw. |
7. exp OBSTETRIC LABOR, PREMATURE/ |
8. exp PREMATURE BIRTH/ |
9. ((preterm or pre-term or premature) adj3 (birth* or labo?r or deliver*)).tw. |
10. or/1-9 |
11. Socioeconomics/ |
12. Cost benefit analysis/ |
13. Cost effectiveness analysis/ |
14. Cost of illness/ |
15. Cost control/ |
16. Economic aspect/ |
17. Financial management/ |
18. Health care cost/ |
19. Health care financing/ |
20. Health economics/ |
21. Hospital cost/ |
22. (fiscal or financial or finance or funding).tw. |
23. Cost minimization analysis/ |
24. (cost adj estimate$).mp. |
25. (cost adj variable$).mp. |
26. (unit adj cost$).mp. |
27. or/11-26 |
28. Developing Country.sh. |
29. (Africa or Asia or Caribbean or West Indies or South America or Latin America or Central America).hw,ti,ab,cp. |
30. (Afghanistan or Albania or Algeria or Angola or Antigua or Barbuda or Argentina or Armenia or Armenian or Aruba or Azerbaijan or Bahrain or Bangladesh or Barbados or Benin or Byelarus or Byelorussian or Belarus or Belorussian or Belorussia or Belize or Bhutan or Bolivia or Bosnia or Herzegovina or Hercegovina or Botswana or Brasil or Brazil or Bulgaria or Burkina Faso or Burkina Fasso or Upper Volta or Burundi or Urundi or Cambodia or Khmer Republic or Kampuchea or Cameroon or Cameroons or Cameron or Camerons or Cape Verde or Central African Republic or Chad or Chile or China or Colombia or Comoros or Comoro Islands or Comores or Mayotte or Congo or Zaire or Costa Rica or Cote d'Ivoire or Ivory Coast or Croatia or Cuba or Cyprus or Czechoslovakia or Czech Republic or Slovakia or Slovak Republic or Djibouti or French Somaliland or Dominica or Dominican Republic or East Timor or East Timur or Timor Leste or Ecuador or Egypt or United Arab Republic or El Salvador or Eritrea or Estonia or Ethiopia or Fiji or Gabon or Gabonese Republic or Gambia or Gaza or Georgia Republic or Georgian Republic or Ghana or Gold Coast or Greece or Grenada or Guatemala or Guinea or Guam or Guiana or Guyana or Haiti or Honduras or Hungary or India or Maldives or Indonesia or Iran or Iraq or Isle of Man or Jamaica or Jordan or Kazakhstan or Kazakh or Kenya or Kiribati or Korea or Kosovo or Kyrgyzstan or Kirghizia or Kyrgyz Republic or Kirghiz or Kirgizstan or Lao PDR or Laos or Latvia or Lebanon or Lesotho or Basutoland or Liberia or Libya or Lithuania or Macedonia or Madagascar or Malagasy Republic or Malaysia or Malaya or Malay or Sabah or Sarawak or Malawi or Nyasaland or Mali or Malta or Marshall Islands or Mauritania or Mauritius or Agalega Islands or Mexico or Micronesia or Middle East or Moldova or Moldovia or Moldovian or Mongolia or Montenegro or Morocco or Ifni or Mozambique or Myanmar or Myanma or Burma or Namibia or Nepal or Netherlands Antilles or New Caledonia or Nicaragua or Niger or Nigeria or Northern Mariana Islands or Oman or Muscat or Pakistan or Palau or Palestine or Panama or Paraguay or Peru or Philippines or Philipines or Phillipines or Phillippines or Poland or Portugal or Puerto Rico or Romania or Rumania or Roumania or Russia or Russian or Rwanda or Ruanda or Saint Kitts or St Kitts or Nevis or Saint Lucia or St Lucia or Saint Vincent or St Vincent or Grenadines or Samoa or Samoan Islands or Navigator Island or Navigator Islands or Sao Tome or Saudi Arabia or Senegal or Serbia or Montenegro or Seychelles or Sierra Leone or Slovenia or Sri Lanka or Ceylon or Solomon Islands or Somalia or South Africa or Sudan or Suriname or Surinam or Swaziland or Syria or Tajikistan or Tadzhikistan or Tadjikistan or Tadzhik or Tanzania or Thailand or Togo or Togolese Republic or Tonga or Trinidad or Tobago or Tunisia or Turkey or Turkmenistan or Turkmen or Uganda or Ukraine or Uruguay or USSR or Soviet Union or Union of Soviet Socialist Republics or Uzbekistan or Uzbek or Vanuatu or New Hebrides or Venezuela or Vietnam or Viet Nam or West Bank or Yemen or Yugoslavia or Zambia or Zimbabwe or Rhodesia).hw,ti,ab,cp. |
31. ((developing or less* developed or under developed or underdeveloped or middle income or low* income or underserved or underserved or deprived or poor*) adj (countr* or nation? or population? or world)).ti,ab. |
32. ((developing or less* developed or under developed or underdeveloped or middle income or low* income) adj (economy or economies)).ti,ab. |
33. (low* adj (gdp or gnp or gross domestic or gross national)).ti,ab. |
34. (low adj3 middle adj3 countr*).ti,ab. |
35. (lmic or lmics or third world or lami countr*).ti,ab. |
36. transitional countr*.ti,ab. |
37. or/28-36 |
38. 10 and 27 and 37 |
See PDF doc for References